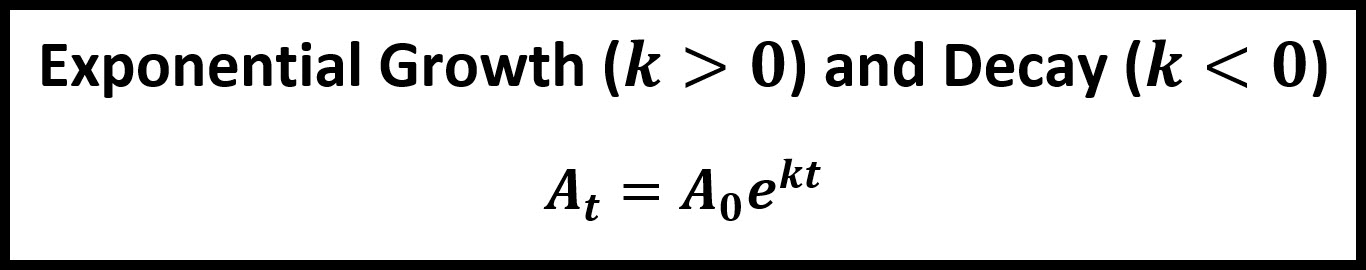Notes
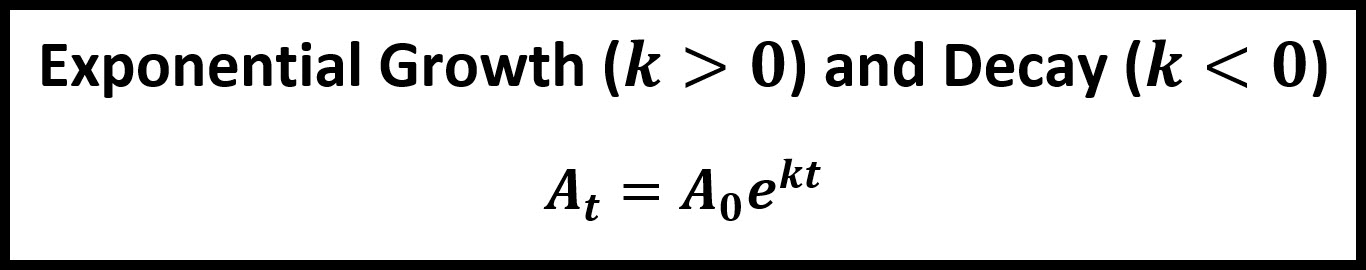
Questions & Videos
A specific radioactive isotope has a half-life of 12,000 years.
\(\textbf{1)}\) What is the decay constant k?
The answer is k\(\approx-.0000577623\)
\(\,\,\,\,\,\,A_t=A_0e^{kt}\)
\(\,\,\,\,\,\,.5=1e^{k12{,}000}\)
\(\,\,\,\,\,\,\ln{.5}=1\ln{e^{k12{,}000}}\)
\(\,\,\,\,\,\,\ln{.5}=k12{,}000\)
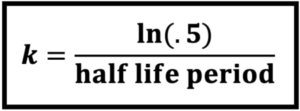
\(\,\,\,\,\,\,\displaystyle\frac{\ln{.5}}{12{,}000}=k\)
\(\,\,\,\,\,\,\)The answer is k\(\approx-.0000577623\)

\(\textbf{2)}\) How long will it take 5 grams to decay to 1 gram?
The answer is \(27{,}863.14\) years
\(\,\,\,\,\,\,A_t=A_0e^{kt}\)
\(\,\,\,\,\,\,1=5e^{-.0000577623t}\)
\(\,\,\,\,\,\,\frac{1}{5}=e^{-.0000577623t}\)
\(\,\,\,\,\,\,\ln{\frac{1}{5}}=\ln{e^{-.0000577623t}}\)
\(\,\,\,\,\,\,\ln{.2}=-.0000577623t\)
\(\,\,\,\,\,\,\displaystyle\frac{\ln{.2}}{-.0000577623}=t\)
\(\,\,\,\,\,\,27{,}863.14\approx t\)
\(\,\,\,\,\,\,\)The answer is \(27{,}863.14\) years

\(\textbf{3)}\) How much of a 30 gram sample will remain after 20,000 years?
The answer is \(9.449\) grams
\(\,\,\,\,\,\,A_t=A_0e^{kt}\)
\(\,\,\,\,\,\,A_{20{,}000}=30e^{-.0000577623 \cdot (20{,}000)}\)
\(\,\,\,\,\,\,A_{20{,}000}=30e^{-1.155246}\)
\(\,\,\,\,\,\,A_{20{,}000}=30 \cdot (0.31498)\)
\(\,\,\,\,\,\,A_{20{,}000}=9.4494\)
\(\,\,\,\,\,\,\)The answer is \(9.449\) grams
\(\textbf{4)}\) What is the half-life of a substance with a decay constant \(k \approx -0.0001\)?
The answer is \(6{,}931.47\) years
\(\,\,\,\,\,\,t_{\frac{1}{2}} = \displaystyle\frac{\ln{2}}{|k|}\)
\(\,\,\,\,\,\,t_{\frac{1}{2}} = \displaystyle\frac{\ln{2}}{0.0001}\)
\(\,\,\,\,\,\,t_{\frac{1}{2}} \approx 6{,}931.47\)
\(\,\,\,\,\,\,\)The answer is \(6{,}931.47\) years
\(\textbf{5)}\) If a 10-gram sample decays to 3 grams in 5,000 years, what is the decay constant \(k\)?
The answer is \(k \approx -0.00024079\)
\(\,\,\,\,\,\,A_t = A_0e^{kt}\) \(\,\,\,\,\,\,3 = 10e^{k(5{,}000)}\)
\(\,\,\,\,\,\,\displaystyle\frac{3}{10} = e^{k(5{,}000)}\)
\(\,\,\,\,\,\,\ln{\displaystyle\frac{3}{10}} = k(5{,}000)\)
\(\,\,\,\,\,\,k = \displaystyle\frac{\ln{\displaystyle\frac{3}{10}}}{5{,}000}\)
\(\,\,\,\,\,\,k \approx -0.0002231\)
\(\,\,\,\,\,\,\)The answer is \(k \approx -0.00024079\)
\(\textbf{6)}\) How long will it take for a 50-gram sample to decay to 12.5 grams with a decay constant \(k = -0.0001\)?
The answer is \(13{,}862.94\) years
\(\,\,\,\,\,\,A_t = A_0e^{kt}\)
\(\,\,\,\,\,\,12.5 = 50e^{-0.0001t}\)
\(\,\,\,\,\,\,\displaystyle\frac{12.5}{50} = e^{-0.0001t}\)
\(\,\,\,\,\,\,\ln{\displaystyle\frac{12.5}{50}} = -0.0001t\)
\(\,\,\,\,\,\,t = \displaystyle\frac{\ln{\displaystyle\frac{12.5}{50}}}{-0.0001}\)
\(\,\,\,\,\,\,t \approx 13{,}863.14\)
\(\,\,\,\,\,\,\)The answer is \(13{,}862.94\) years
\(\textbf{7)}\) A radioactive substance starts at 100 grams and decays to 25 grams in 10,000 years. What is the half-life?
The answer is \(5{,}000\) years
\(\,\,\,\,\,\,\text{After 1 half-life: } 100 \to 50 \, \text{grams}\)
\(\,\,\,\,\,\,\text{After 2 half-lives: } 50 \to 25 \, \text{grams}\)
\(\,\,\,\,\,\,\text{Time for 2 half-lives: } 10{,}000 \, \text{years}\)
\(\,\,\,\,\,\,\text{Each half-life is } \frac{10{,}000}{2} = 5{,}000 \, \text{years}\)
\(\,\,\,\,\,\,\)The answer is \(5{,}000\) years
\(\textbf{8)}\) How much of a 200-gram sample remains after 15,000 years, if \(k = -0.0000462\)?
The answer is about\(100\) grams
\(\,\,\,\,\,\,A_t = A_0e^{kt}\)
\(\,\,\,\,\,\,A_{15{,}000} = 200e^{-0.0000462 \cdot (15{,}000)}\)
\(\,\,\,\,\,\,A_{15{,}000} = 200e^{-0.693}\)
\(\,\,\,\,\,\,A_{15{,}000} = 200 \cdot (0.500074)\)
\(\,\,\,\,\,\,A_{15{,}000} \approx 100\)
\(\,\,\,\,\,\,\)The answer is about \(100\) grams
\(\textbf{9)}\) What is the remaining amount of a substance if it starts at 80 grams and decays for 30,000 years with a half-life of 15,000 years?
The answer is \(20\) grams
\(\,\,\,\,\,\,\text{Each half-life reduces the sample by half.}\)
\(\,\,\,\,\,\,\text{After 1 half-life (15,000 years): } 80 \to 40 \, \text{grams}\)
\(\,\,\,\,\,\,\text{After 2 half-lives (30,000 years): } 40 \to 20 \, \text{grams}\)
\(\,\,\,\,\,\,\)The remaining amount after 30,000 years is \(20\) grams
\(\textbf{10)}\) A sample has a half-life of 10,000 years. How long will it take to decay to \(1/8\) of its original mass?
The answer is \(30{,}000\) years
\(\,\,\,\,\,\,\text{Each half-life reduces the sample by half.}\)
\(\,\,\,\,\,\,\text{After 1 half-life: } \frac{1}{2}\)
\(\,\,\,\,\,\,\text{After 2 half-lives: } \frac{1}{4}\)
\(\,\,\,\,\,\,\text{After 3 half-lives: } \frac{1}{8}\)
\(\,\,\,\,\,\,t = 3 \cdot 10{,}000\)
\(\,\,\,\,\,\,t = 30{,}000\)
\(\,\,\,\,\,\,\)The answer is \(30{,}000\) years
See Related Pages\(\)